Fracture Risk Following SCI
The vast majority of current evidence supports the importance of addressing fracture risk after SCI since there is a higher incidence of fragility fractures in this population (Table 1). The majority of fragility fractures occur following transfers or activities that involve minimal or no trauma (Comarr et al. 1962; Ragnarsson & Sell; 1981; Freehafer 1995; Akhigbe et. al 2015) where the distal femur and proximal tibia (knee region) are most at risk.
Recent findings from review studies in veterans (n=12,162) with SCI have found that 82.6% of all fractures were at the tibia/fibula, femur or hip (Fig 1). Further, individuals with SCI were less likely to receive surgical intervention, than people without SCI, although those with SCI who have surgery did not have increased mortality or adverse event rates (Bishop et. al, 2013; Bethel et. al, 2015). Delayed fracture union is common after SCI (Grassner et al. 2017). Following a fracture there is a five-year increased risk of mortality (Pelletier et al. 2014; Carbone et al. 2014).
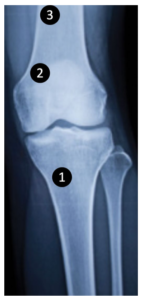 Figure 1. Many of the fractures that individuals with SCI sustain occur in the region of the metaphysis and epiphysis. Source: http://sci.washington.edu/info/forums/reports/osteoporosis.asp#dx
Figure 1. Many of the fractures that individuals with SCI sustain occur in the region of the metaphysis and epiphysis. Source: http://sci.washington.edu/info/forums/reports/osteoporosis.asp#dx
Risk factors for fragility fracture after SCI include:
- Sex
- Age at injury
- Time Post-Injury
- Type of impairment
- Low BMI
- Low knee region BMD, and
- Use of anticonvulsants, heparin, or opioid analgesics.
Women are at greater risk compared to men (Vestergaard et al. 1998; Lazo et al. 2001; Nelson et al. 2003; Garland et al. 2004). Increasing age and longer TPI (Frisbie 1997; McKinley et al. 1999; Garland et al. 2004; Garland et al. 2005) increases fracture risk which rises significantly at 10 years post-injury. Further, people with paraplegia have more fractures (Frisbie 1997) and those with complete injuries have greater bone mass loss compared with those with incomplete injuries (Garland et al. 2004; Garland et al. 2005).
A number of concurrent medications a patient is taking can also decrease or substantially increase fracture risk. These include but are not limited to: heparin, benzodiazepines, anticonvulsants, proton pump inhibitors, selective serotonin reuptake inhibitors and opioid analgesics. In a large retrospective cohort study of men with chronic SCI (n=6969, ≥ 2 years post-injury), the use of thiazide-type diuretics was associated with a 25% reduction in the risk of lower extremity fragility fractures (Carbone et al. 2013c). In contrast, the use of heparin (HR 1.48, CI 1.20-1.83), opioid analgesics (HR 1.80, CI 1.57-2.06), or anticonvulsants (HR 1.35, CI 1.18-1.54), especially the benzodiazepine sub-class (HR 1.45, CI 1.27-1.65), was associated with an increased risk of lower extremity fragility fractures in men with chronic SCI (≥ 2 years post-injury) (Carbone et al. 2013a, 2013b). Men with chronic SCI are at a slightly increased risk of lower extremity fragility fractures when exposed to proton pump inhibitors (HR 1.08, CI 0.93-1.25), selective serotonin reuptake inhibitors (HR 1.05, CI 0.90-1.23), or thiazolidinediones (HR 1.04, CI 0.68-1.61) (Carbone et al. 2013a, 2013b). However, these drugs and a prior history of fragility fracture or a history of fracture in a parent are known risk factors for the development of osteoporosis in the general population, and should, therefore, be considered when assessing fracture risk in SCI patients.
Fracture thresholds are values below which fragility fractures begin to occur, whereas fracture breakpoints are values below which the majority of fractures occur (Garland et al. 2005). Knee region areal BMD (aBMD) and volumetric (vBMD) thresholds for fracture and breakpoint have been identified (Mazess 1990; Eser et al. 2005; Garland et al. 2005). BMD thresholds are described on Table 2.
We recommend documenting your patient’s fracture risk by completing the risk factor profile checklist (Craven et al. 2008; Craven et al. 2009). We propose that the presence of ≥ 3 risk factors implies a moderate fracture risk, and ≥ 5 risk factors implies a high fracture risk (Table 3).
Gap: Fracture Management After SCI |
|---|
|
Source of evidence |
|
Recognizing a fracture
|
|
Management |
Bethel et al. 2015 compared results of incident fracture treatment (surgical vs. non- surgical) among male veterans with chronic SCI. The study comprised 1,979 incident fractures that occurred among 1281 veterans over 6 years. The majority of fractures occurred in lower extremities (~83%), and the majority of these fractures were treated nonsurgically (~90%). The authors reported that there was a significant difference in the level of injury and fracture treatment modality, surgery treatment being used more among individuals with paraplegia (p = 0.04). However, there were no significant fracture treatment-related differences in mortality rates.
