FES Cycle Ergometry
Discussion
There are mixed results for bone outcomes after FES-cycling. Three studies reported a regional increase in BMD (Mohr et al. 1997; Chen et al. 2005; Frotzler et al. 2008) at the distal femur or proximal tibia, while there was no significant within-participant BMD change at the hip in three pre-post studies (Pacy et al. 1988; Leeds et al. 1990; and BeDell et al. 1996). The FES-cycling studies which reported a positive effect on bone parameters used protocols that were at least 3 sessions/week for 6 months and increased bone parameters over areas directly affected by stimulated muscles (e.g., quads, distal femur and proximal tibia). Although one study showed that FES-cycling intervention needed to be maintained or bone gains were lost (Chen et al. 2005).
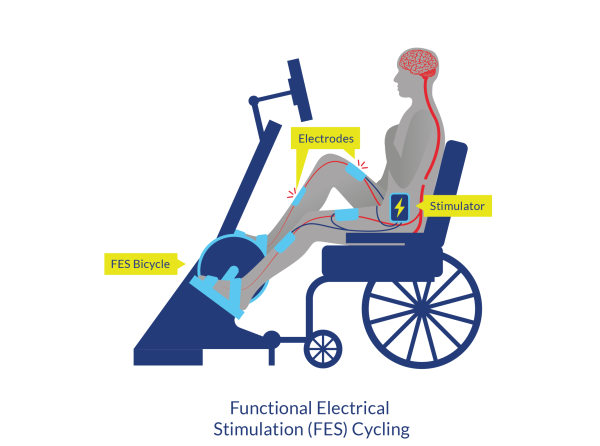
Figure 4. Functional Electrical Stimulation (FES) Cycling
Frotzler et al. (2008) found BMD and BMC were preserved at the distal sites for some participants at 12 months. Hammond et al. (2014) suggested that FES-cycling may reduce the prevalence of osteoporosis in people with SCI compared to people with SCI that do not use FES-cycling; however, no minimal FES requirements (i.e. intensity, duration or frequency) were provided. Craven et. al, (2017) and Johnston et al. (2016) however, showed no difference in BMD after 4-6 months of FES-walking and FES-cycling, respectively. In summary, longitudinal FES-cycling shows promise as an effective treatment for regional BMD maintenance (around the knee, where fracture risk is highest). The limited availability of FES-cycling for home or longitudinal use may limit its generalizability outside a clinical trial scenario.
Conclusions
There is level I evidence that FES-walking did not elicit significant changes in bone biomarkers or BMD after 4 months of training (Craven et al. 2017)
Johnston et al. (2016) showed that 6 months of FES-cycling did not produce significant changes in bone architecture.
Hammond et al. 2014 suggested a decrease in the prevalence of osteoporosis after FES-cycling.
There is level 4 evidence (Mohr et al. 1997; Chen et al. 2005; Frotzler et al. 2008) that FES-cycling increased regional lower extremity BMD over areas stimulated.
