Cough Assist and Secretion Removal
People with SCI are at risk for retention of secretions because of an increased prevalence of pneumonia compounded by lower expiratory flows during cough, which is greatest during the acute phase after SCI. Increased prevalence of RI, although decreased during the rehabilitation phase of recovery, is still higher in people with SCI compared to age-matched healthy people, and secretion retention is a major contributor to respiratory illness in people with SCI (Reid et al. 2010). Reduction in expiratory flows during cough is related to the higher levels of SCI. Of considerable surprise, several devices that have been shown to be effective in people with other chronic respiratory conditions have not been evaluated in people with SCI.
Discussion
Respiratory complications are a primary cause of morbidity and mortality in people with SCI. Though some studies have examined the effectiveness of secretion removal techniques, there are discrepancies in how standard pulmonary function is measured (Kang et al. 2006). Limited evidence supports the postulate that improving inspiratory muscle strength (Kang et al. 2006) in addition to expiratory muscle force (Estenne et al. 2000) are important to maximize expiratory flows during cough. IMT (Van Houtte et al. 2008), ES of the expiratory muscles (Linder 1993; Estenne et al. 2000; DiMarco et al. 2009, Butler et al. 2011), and MIE (the application of positive pressure to the airway, then shifting to negative pressure to produce an expiratory flow simulating a cough) as an adjunct to manual respiratory kinesitherapy (Pillastrini et al. 2006) are three potential therapies that can maximize the force produced by the inspiratory and expiratory muscles to increase expiratory flows during cough. RCTs examining the effectiveness of airway clearance techniques in people after SCI are lacking. RMT (Van Houtte et al. 2008) and MIE (Crew et al. 2010) have been shown to decrease infections and respiratory hospitalizations per year.
The series of pre–post studies (DiMarco et al. 2019; DiMarco et al. 2020; DiMarco et al. 2021; DiMarco et al. 2022) showed that spinal cord stimulation (SCS) was effective in the increase of peak expiratory airflow, airway pressure generation, and expiratory muscle function, with an associated effect of ease in raising secretions and decrease acute RTI. Moreover, when this group compared the clinical outcomes of wire electrodes (which can be placed using minimally invasive techniques and associated reduction in cost, surgical time and overall risk) and disc electrodes, they showed no differences in the increase of secretions between types of electrodes (DiMarco et al. 2022).
GPB can be used to increase lung volumes and assist secretion clearance in patients with high tetraplegia (Pryor 1999) and in ventilator users, GPB can provide security in case of ventilator failure or for brief periods of ventilator-free breathing (Dail et al. 1956). Nygren-Bonnier et al. (2009) showed that a GPB training for 8 weeks could be adopted by most patients (20/25) and provided an increase in most pulmonary function parameters (VC, ERV, FRC, RV, TLC, and alveolar ventilation), an additional insufflation of 28% of their VC; and an improvement of subjective ability to cough and clear secretions. However, the participants had occasional symptoms during glossopharyngeal insufflation (GI) such as dizziness, increased tightening of the chest, localized numbness, and fainting (syncope). The more recent pre–post study of Nygren-Bonnier et al. (2018), compared the acute effects of GI and showed greater increases in the group with SCI in TLC, VC, mouth airway pressure, HR and in the decrease in MAP, though there were no difference between groups in supine position.
Further study in SCI should examine the effectiveness of hand-held devices that facilitate airway clearance, such as those that apply continuous (Peripep®) or oscillating positive expiratory pressure (Flutter). Of equal concern is to evaluate the comfort and preference of airway clearance techniques that are readily adhered to and performed by people with SCI. Some evidence supports the effectiveness of these positive expiratory pressure devices and other secretion removal techniques such as autogenic drainage in people with cystic fibrosis and other chronic respiratory diseases; however, the evidence to date is somewhat equivocal (Hess 2001; Reid & Chung 2004).
Gap: SCI Evidence on the Use of LVR (Lung Volume Recruitment) and Assisted Cough for Secretion Management |
|---|
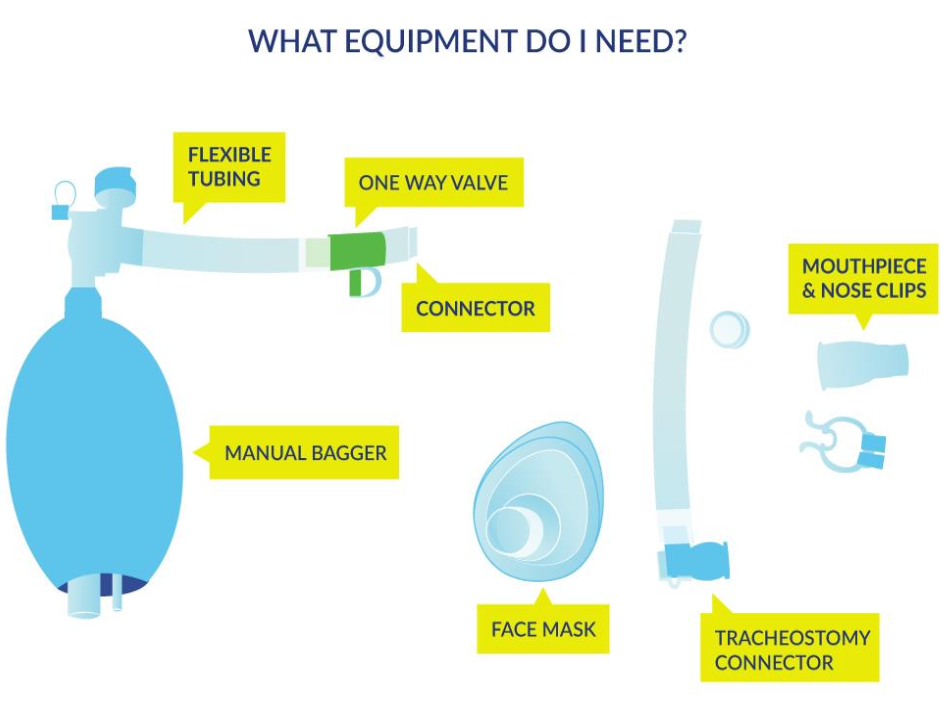
Source of evidenceWe found one study using LVR for people with SCI (see above Molgat-Seon et al. 2017). However, there is a large body of evidence from other populations with neurological respiratory impairment and cough impairment, predominantly Duchennes Muscular Dystrophy, Amyotrophic Lateral Sclerosis and Multiple Sclerosis. There are a variety of LVR techniques possible: using a LVR resuscitation bag, using a MIE machine or using the Ventilator for individuals already using one. |
Recognizing risk of impaired secretion clearancePatients with SCI commonly develop restrictive lung disorders as a result of their decreased respiratory muscle strength, reduced VC, ineffective cough and reduced lung and chest wall compliance. These acute and chronic chest changes place people with SCI at risk for cardiorespiratory complications such as atelectasis, secretion retention and recurrent chest infections. Mechanical in-exsufflation (e.g., cough assist machines), lung volume augmentation techniques (e.g., breath-stacking) and manual assisted cough techniques are recommended as best practice for managing acute and chronic cardiorespiratory conditions in people with SCI. Persons with a PCF of less than 270 L/min are at risk for secretion retention and need manual or mechanical assistance to avoid serious complications or health risks. |
Management:Assisted cough: This is a manual technique used to increase expiratory pressure. It is used to compensate for the decreased intra-abdominal pressure that can be present with certain levels of SCI. According to the Australian and New Zealand clinical practice guidelines for physiotherapy interventions for the respiratory management of people with SCI (Tranter et al. 2025), there is a strong consensus-based opinion statement that this technique should be provided to improve secretion clearance in people with SCI who have abdominal muscle weakness or paralysis and an ineffective cough. Pressure is applied in the direction of the costal and abdominal areas during expiration. It can be done in lying or sitting PRN depending on need. Appropriate communication and timing are required to ensure that the manual thrust is done just at or prior to expiration. There are some precautions and contraindications mostly related to abdominal trauma, fractures etc.
|
Conclusion
Secretion removal techniques are common practice in people with SCI and yet there is predominantly level 4 evidence to support the use of some airway clearance techniques to facilitate secretion removal in this population.
There is level 2 evidence (from two RCTs: Pillastrini et al. 2006; Jeong & Yoo 2015) in support of MIE coupled with manual chest therapy kinesitherapy techniques.
There is level 4 evidence (from four pre-post studies: DiMarco et al. 2019; DiMarco et al. 2020; DiMarco et al. 2021; DiMarco et al. 2022) that SCS improves expiratory and inspiratory muscle function, peak expiratory airflow, airway pressure generation; with a subsequent increment of ease in raising secretions and descend of acute RTI.
There is level 4 evidence (from one pre–post study: Nygren-Bonnier et al. 2009) that a GPB training period of 8 weeks provides beneficial effects on respiratory parameters (as measured by VC, ERV, FRC, RV, TLC, and alveolar ventilation) and in subjective ability to cough and clear secretions in patients with cervical SCI.
There is no evidence in support of one airway clearance technique over another, and there are no criteria available to indicate when to implement the various airway clearance techniques.
There is a need to determine the most efficient and effective techniques that are comfortable and readily adhered to for people with SCI in order to facilitate airway clearance, improve their QOL, and decrease health care costs.
 Manual assisted coughing
Manual assisted coughing